Market Definition
The market comprises diagnostic instruments, consumables, and testing platforms used to determine the effectiveness of antimicrobial agents against specific pathogens. These solutions evaluate microbial response to antibiotics or antifungals through phenotypic, genotypic, and automated methods, generating data such as resistance profiles and minimum inhibitory concentrations.
AST systems convert laboratory findings into clinically actionable insights that support appropriate therapy selection, antimicrobial stewardship, and infection control. They play a critical role in infection diagnosis, antimicrobial stewardship, and resistance surveillance across hospitals, clinical laboratories, diagnostic centers, pharmaceutical research facilities, and public health institutions.
Antimicrobial Susceptibility Testing Market Overview
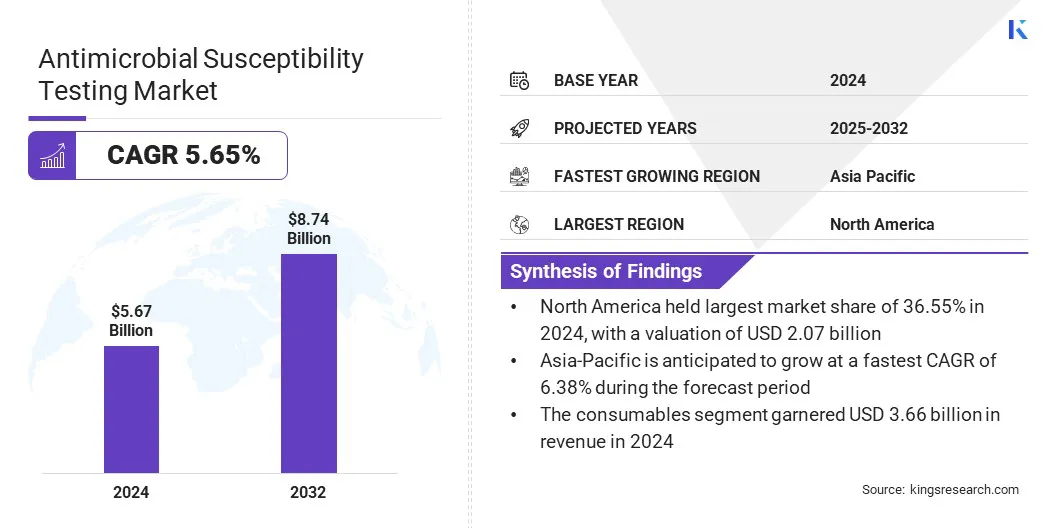
The global antimicrobial susceptibility testing market size was valued at USD 5.67 billion in 2024 and is projected to grow from USD 5.95 billion in 2025 to USD 8.74 billion by 2032, exhibiting a CAGR of 5.65% over the forecast period.
The market is expanding due to increasing antimicrobial resistance and the growing need for timely, accurate therapy selection. Market growth is supported by rising infection prevalence, expanding antimicrobial stewardship programs (ASPs), and wider adoption of automated and rapid AST technologies across healthcare settings.
Key Market Highlights:
- The antimicrobial susceptibility testing industry size was recorded at USD 5.67 billion in 2024.
- The market is projected to grow at a CAGR of 5.65% from 2024 to 2032.
- North America held a market share of 36.55% in 2024, with a valuation of USD 2.07 billion.
- The consumables segment garnered USD 3.66 billion in revenue in 2024.
- The broth dilution segment is expected to reach USD 3.99 billion by 2032.
- The clinical diagnostics segment is expected to register a market share of 56.80% by 2032.
- The hospitals & clinical laboratories segment registered a market share of 56.43% in 2024.
- Asia Pacific is anticipated to grow at a CAGR of 6.38% during the forecast period.
Major companies operating in the antimicrobial susceptibility testing market are Accelerate Diagnostics, Inc., Alifax S.r.l., Beckman Coulter, Inc., BD, Bio-Rad Laboratories, Inc, BIOMÉRIEUX, Bruker, Cepheid., Creative Diagnostics, F. Hoffmann-La Roche, Hain Lifescience GmbH, Hardy Diagnostics, HiMedia Laboratories., Liofilchem S.r.l., and Merck KGaA.
Market growth is driven by the increasing need for accurate interpretation of complex antimicrobial susceptibility data to support effective clinical decision-making. Rising antimicrobial resistance has increased the clinical importance of combining rapid pathogen identification with reliable susceptibility profiling to guide targeted therapy.
Healthcare providers are adopting advanced AST systems that integrate automated testing with decision-support and informatics capabilities to reduce interpretation variability and improve diagnostic confidence.
Leading companies such as BD, BIOMÉRIEUX, Accelerate Diagnostics, Inc., and others continue to enhance AST platforms with integrated analytics and workflow support to improve data usability and standardization, reinforcing broader adoption of antimicrobial susceptibility testing solutions across clinical laboratories.
- For instance, in April 2025, BD (Becton, Dickinson and Company) announced the clearance of its BD Phoenix M50 and BDXpert System integrated with the BD Synapsys Informatics Solution. The platform leverages rapid pathogen identification and antimicrobial susceptibility testing algorithms to support accurate interpretation of complex diagnostic data, improving reliability in the detection and management of antimicrobial resistance.
What is driving the adoption of antimicrobial susceptibility testing amid rising antimicrobial resistance?
The market growth is driven by the rising global burden of antimicrobial resistance, which has increased the need for accurate and timely identification of effective therapies. Hospitals and clinical laboratories increasingly rely on antimicrobial susceptibility testing to guide targeted treatment decisions, reduce inappropriate antibiotic use, and comply with antimicrobial stewardship requirements.
Increasing infection rates, extended hospital stays associated with resistant pathogens, and stronger regulatory emphasis on resistance monitoring are further reinforcing routine AST adoption across healthcare systems.
- According to the Centers for Disease Control and Prevention (CDC), antimicrobial resistance represents an urgent global public health threat, contributing to at least 1.27 million deaths worldwide. In the U.S., more than 2.8 million antimicrobial-resistant infections occur each year.
What strategies are market participants adopting to overcome the high cost of antimicrobial susceptibility testing systems?
A key challenge restraining the growth of the antimicrobial susceptibility testing market is the high cost and operational complexity associated with implementing and maintaining advanced AST systems.
Deploying automated and rapid susceptibility platforms requires significant capital investment, laboratory infrastructure upgrades, and skilled microbiology personnel. In addition, variability in testing standards, validation requirements, and integration with laboratory information systems increases workflow complexity for clinical laboratories. These factors may limit adoption, particularly among smaller hospitals, diagnostic centers, and resource-constrained healthcare settings.
To address this challenge, market participants are focusing on modular system designs, automation-friendly workflows, and standardized testing panels, while expanding training programs, technical support services, and partnerships with healthcare providers to improve accessibility and support wider AST adoption.
How is the integration of automated antimicrobial susceptibility testing technologies reshaping laboratory workflows?
A key trend influencing the antimicrobial susceptibility testing market is the increasing integration of rapid and automated AST technologies, into routine laboratory workflows. Clinical laboratories are moving beyond manual and standalone testing methods toward systems embedded within laboratory information systems and microbiology automation platforms to support seamless susceptibility analysis and reporting.
This integration enables faster result generation, standardized interpretation, and improved data connectivity across diagnostic processes. Embedding automated and digitally enabled AST solutions within existing laboratory environments improves operational efficiency, reduces turnaround times, and supports consistent antimicrobial stewardship practices, thereby accelerating adoption across healthcare settings and driving sustained market demand.
- For instance, in January 2025, Rapid Infection Diagnostics Inc. (RID) announced the launch of BSIDx, a novel microbiology testing system for bloodstream infections. The platform enables automated pathogen identification and antimicrobial susceptibility testing directly from positive blood cultures, delivering results in under five hours, thereby supporting faster clinical decision-making and timely initiation of targeted therapy.
Antimicrobial Susceptibility Testing Market Report Snapshot
|
Segmentation
|
Details
|
|
By Product Type
|
Instruments, Consumables
|
|
By Method
|
Disk Diffusion Method, Broth Dilution, Agar Dilution, Others
|
|
By Application
|
Clinical Diagnostics, Drug Discovery & Development, Epidemiology & Surveillance, Others
|
|
By End User
|
Hospitals & Clinical Laboratories, Diagnostic Centers, Pharmaceutical & Biotechnology Companies, Others
|
|
By Region
|
North America: U.S., Canada, Mexico
|
|
Europe: France, UK, Spain, Germany, Italy, Russia, Rest of Europe
|
|
Asia-Pacific: China, Japan, India, Australia, ASEAN, South Korea, Rest of Asia-Pacific
|
|
Middle East & Africa: Turkey, U.A.E., Saudi Arabia, South Africa, Rest of Middle East & Africa
|
|
South America: Brazil, Argentina, Rest of South America
|
Market Segmentation:
- By Product Type (Instruments and Consumables): The consumables segment earned USD 3.66 billion in 2024, due to its recurring usage in routine testing and rising diagnostic volumes across hospitals and laboratories.
- By Method (Disk Diffusion Method, Broth Dilution, Agar Dilution, and Others): The broth dilution held 43.21% of the market in 2024, driven by its high accuracy in determining minimum inhibitory concentrations and strong integration with automated AST systems.
- By Application (Clinical Diagnostics, Drug Discovery & Development, Epidemiology & Surveillance, and Others): The clinical diagnostics segment is projected to reach USD 4.96 billion by 2032, owing to increasing antimicrobial-resistant infections and growing reliance on AST for targeted therapy selection.
- By End Use (Hospitals & Clinical Laboratories, Diagnostic Centers, Pharmaceutical & Biotechnology Companies, and Others): The hospitals & clinical laboratories segment is projected to reach USD 4.82 billion by 2032, fueled by high patient testing volumes and widespread implementation of antimicrobial stewardship programs.
What is the market scenario in North America and Asia-Pacific region?
Based on region, the market has been classified into North America, Europe, Asia Pacific, Middle East & Africa, and South America.

North America antimicrobial susceptibility testing market share stood around 36.55% in 2024 in the global market, with a valuation of USD 2.07 billion. This dominance is attributed to the advanced healthcare infrastructure, high diagnostic testing volumes, and strong regulatory emphasis on antimicrobial stewardship.
High prevalence of healthcare-associated infections is prompting healthcare providers in this region to implement routine antimicrobial susceptibility testing as a standard component of infection diagnosis and treatment optimization.
In addition, government initiatives, surveillance programs, and guidelines issued by public health agencies further reinforce demand for accurate and timely susceptibility testing. High adoption of automated and rapid AST platforms, along with early uptake of innovative phenotypic and molecular technologies is further driving the market growth across North America.
- According to the Centers for Disease Control and Prevention (CDC), approximately 1 in 31 hospital patients in the U.S. has at least one healthcare-associated infection, highlighting the critical need for effective infection control and antimicrobial susceptibility testing in clinical settings.
Asia-Pacific is poised for significant growth at a robust CAGR of 6.38% over the forecast period, driven by its large patient population, high incidence of infectious diseases, and increasing exposure to antibiotic misuse across both hospital and community settings.
The region experiences elevated rates of drug-resistant infections due to the widespread misuse and over prescription of antibiotics, which accelerates microbial resistance development, particularly in densely populated and rapidly urbanizing areas, which is intensifying the need for routine susceptibility testing. Market expansion is further fueled by the scaling of public hospital networks, the expansion of diagnostic laboratories, and the gradual standardization of microbiology practices.
Regulatory Frameworks
- In the U.S., antimicrobial susceptibility testing is regulated by the U.S. Food and Drug Administration (FDA) under in vitro diagnostic (IVD) medical device regulations. AST instruments, panels, and automated systems require FDA clearance or approval to ensure analytical accuracy, clinical validity, and safety.
- In the European Union, AST products are regulated under the In Vitro Diagnostic Regulation (IVDR), which imposes stricter requirements for clinical evidence, performance evaluation, and post-market surveillance. The European Medicines Agency (EMA) and national competent authorities oversee compliance, while EUCAST (European Committee on Antimicrobial Susceptibility Testing) establishes standardized susceptibility breakpoints and testing methodologies used across member states.
- In APAC, China regulates AST devices through the National Medical Products Administration (NMPA), with increasing emphasis on performance validation and quality standards.
- Globally, the World Health Organization (WHO) plays a key role in setting global standards for antimicrobial resistance surveillance and susceptibility testing. WHO promotes harmonized AST methodologies, global data sharing, and stewardship frameworks to support consistent resistance monitoring and responsible antimicrobial use across healthcare systems worldwide.
Competitive Landscape
Leading players in the antimicrobial susceptibility testing industry are focusing on advancing automation, speed, and clinical integration to strengthen their competitive positioning.
Major diagnostic companies are launching rapid and fully automated AST platforms that deliver faster results directly from positive cultures, addressing critical care and sepsis management needs. Several players are integrating AST systems with laboratory information systems and digital microbiology workflows to improve standardization and data connectivity.
- In June 2023, Sysmex Europe launched the world’s first point-of-care antimicrobial susceptibility testing system capable of generating susceptibility results from urine samples in approximately 30 minutes, compared with the conventional 48–72-hour timeframe. The system leverages proprietary microfluidic technology to enable rapid bacterial detection within about 15 minutes and automated assessment of antibiotic effectiveness, all within a compact desktop platform designed for near-patient testing environments.
Key Companies in Antimicrobial Susceptibility Testing Market:
- Accelerate Diagnostics, Inc.
- Alifax S.r.l.
- Beckman Coulter, Inc.
- BD
- Bio-Rad Laboratories, Inc
- BIOMÉRIEUX
- Bruker
- Cepheid.
- Creative Diagnostics
- Hoffmann-La Roche
- Hain Lifescience GmbH
- Hardy Diagnostics
- HiMedia Laboratories.
- Liofilchem S.r.l.
- Merck KGaA
Recent Developments
- In March 2025, bioMérieux announced FDA 510(k) clearance for VITEK COMPACT PRO, an advanced system for microorganism identification and antimicrobial susceptibility testing. The platform supports clinical laboratories in diagnosing infectious diseases and managing antimicrobial resistance, while also enabling industrial laboratories to identify contaminants and enhance consumer safety.


