Acute Intermittent Porphyria Market Size
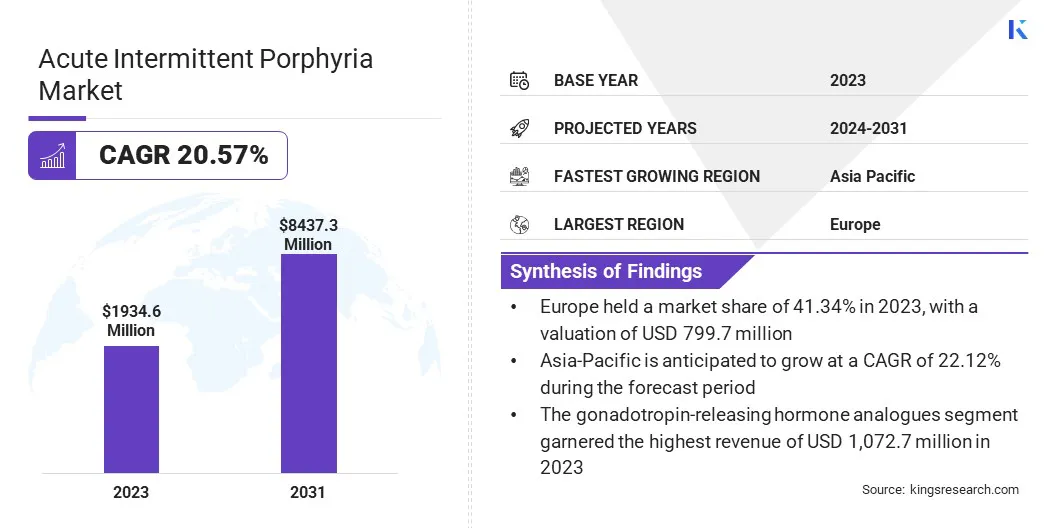
Global Acute Intermittent Porphyria Market size was recorded at USD 1,934.6 million in 2023, which is estimated to be at USD 2,277.5 million in 2024 and projected to reach USD 8,437.3 million by 2031, growing at a CAGR of 20.57% from 2024 to 2031.
In the scope of work, the report includes solutions offered by companies such Alnylam Pharmaceuticals, Inc., Recordati Rare Diseases, Octapharma AG, Clinuvel Pharmaceuticals, Shandong Octagon Chemicals Limited, Glenmark Pharmaceuticals Ltd, Intas Pharmaceutical Ltd., Sanofi, Sun Pharmaceuticals Industries Ltd., Abbott Healthcare Pvt. Ltd. and others.
The growth of the market is driven by increasing awareness and recognition of acute intermittent porphyria among healthcare professionals and the general population. This has led to higher rates of diagnosis and increased demand for specialized diagnostic tools and therapeutic solutions tailored to individual patient needs.
The expansion of the acute intermittent porphyria market is further fueled by ongoing advancements in genetic testing technologies, enabling early and accurate diagnosis, along with the rising prevalence of metabolic disorders.
Additionally, a growing trend toward patient-centric care models emphasizes personalized treatment plans and holistic disease management, thereby enhancing patient outcomes. Opportunities are present in the development of novel therapies and personalized medicine approaches, as well as in the utilization of genetic insights for mutation-specific treatments.
- Regulatory support and guidelines offer opportunities for implementing standardized approaches in both diagnosis and treatment. Moreover, collaboration among healthcare professionals and patient advocacy groups fosters innovation and awareness in the market, providing avenues for improved patient care and market expansion.
Acute Intermittent Porphyria (AIP) is a rare metabolic disorder characterized by a deficiency of the enzyme hydroxymethylbilane synthase (HMBS), also known as porphobilinogen deaminase (PBGD). This enzymatic deficiency leads to the accumulation of toxic porphyrin precursors in the body. However, the majority of individuals with HMBS gene mutations do not typically exhibit symptoms of AIP.
Symptom onset often requires additional factors such as endocrine influences, medication use, alcohol consumption, infections, or dietary changes to trigger. The market encompasses diagnostic tests, therapeutic interventions, and supportive care solutions aimed at addressing the diagnosis, management, and mitigation of symptoms associated with this rare metabolic disorder.
Analyst’s Review
The acute intermittent porphyria market shows considerable growth potential due to increasing healthcare awareness and the rising incidence of metabolic disorders.
Diagnostic firms are increasingly introducing advanced technologies for early and precise AIP diagnosis, thereby ensuring timely intervention and personalized treatment plans. Furthermore, genetic testing advancements are playing a crucial role in facilitating early detection and better management of AIP by identifying individuals at risk before symptoms develop.
Moreover, strategic collaborations between pharmaceutical companies and healthcare providers are essential in advancing AIP management. These partnerships are fostering the development of integrated care pathways and robust patient education programs.
- For instance, in April 2024, the American Porphyria Foundation (APF) partnered with Recordati Rare Diseases, for the National Porphyria Awareness Week (NPAW) initiative, which aimed to raise awareness of porphyria. This initiative played a significant role in enhancing public understanding and support for AIP.
Acute Intermittent Porphyria Market Growth Factors
The growing prevalence of metabolic disorders and diseases significantly influences the acute intermittent porphyria market. As metabolic disorders become increasingly common due to sedentary lifestyles, poor diet, and genetic predispositions, the need for comprehensive diagnostic and treatment solutions is growing. AIP, a rare metabolic disorder, often intersects with other metabolic conditions, thereby necessitating an integrated approach to patient care.
- According to the American Porphyria Foundation, AIP is more common in women, possibly attributable to fluctuations in menstrual cycle hormones. About 3%-5% of affected individuals, predominantly women, experience recurrent attacks.
This rise in metabolic disorders has led to more frequent screening and diagnosis of AIP, thereby boosting demand for specialized diagnostic tools and therapeutic options. The overlap between AIP and other metabolic diseases drives research into innovative treatments and management strategies, thus propelling market growth.
However, the complexity of diagnosis and the often-delayed identification of the condition are expected to pose challenges to market expansion. AIP shares symptoms with various other diseases, which can lead to misdiagnosis or delayed diagnosis, thereby exacerbating the severity of symptoms and hindering effective management.
This challenge underscores the need for improved awareness among healthcare professionals and individuals regarding the signs and symptoms of AIP, as well as the importance of early and accurate diagnosis.
Additionally, advancements in diagnostic technologies, such as genetic testing and biomarker identification, are essential for improving the accuracy and speed of AIP diagnosis. Moreover, fostering interdisciplinary collaboration among healthcare professionals, including specialists in genetics, gastroenterology, and hematology, ensures comprehensive assessment and management of AIP cases, thereby leading to market development.
Acute Intermittent Porphyria Market Trends
The acute intermittent porphyria market is experiencing significant growth, mainly propelled by ongoing advancements in genetic testing technologies. These innovations are enhancing both the accuracy and speed of diagnosing AIP, enabling early detection and timely intervention.
Improved genetic testing allows for precise identification of the specific mutations responsible for AIP, facilitating targeted and personalized treatment approaches. This technological progress reduces the time and cost associated with traditional diagnostic methods, while also minimizing the risk of misdiagnosis, thereby enhancing patient outcomes.
The increasing adoption of these advanced genetic tests by healthcare providers is propelling the demand for specialized diagnostic services. Moreover, pharmaceutical companies are leveraging genetic testing to develop more effective, mutation-specific therapies, thus contributing significantly to market growth.
- The Mount Sinai Genetic Testing Laboratory in New York City has announced the availability of DNA testing for seven types of Porphyria. Notably, it stands as the sole laboratory in the United States to offer comprehensive DNA testing encompassing all these Porphyria. This testing program was developed through funding provided by the American Porphyria Foundation, highlighting the advancements made in genetic testing that facilitate early and accurate diagnosis of these conditions.
Companies operating in the acute intermittent porphyria market are increasingly adopting patient-centric care models, reflecting a significant shift toward personalized treatment and holistic disease management. These models prioritize the specific needs and preferences of patients by offering tailored treatment plans that address both the medical and psychosocial aspects of AIP.
By integrating personalized medicine approaches, healthcare providers are able to deliver more effective and targeted therapies, significantly improving patient outcomes and quality of life.
This trend is further bolstered by advancements in diagnostic technologies and a deeper understanding of the genetic and environmental factors influencing AIP. Healthcare institutions and pharmaceutical companies are investing heavily in comprehensive care programs that include regular monitoring, patient education, and support services, thereby boosting market expansion.
Segmentation Analysis
The global market is segmented based on treatment, end-users, and geography.
By Treatment
Based on treatment, the acute intermittent porphyria market is categorized into gonadotropin-releasing hormone analogues, and prophylactic hematin infusions.
The gonadotropin-releasing hormone analogues segment garnered the highest revenue of USD 1,072.7 million in 2023. This notable segmental growth is largely attributed to their demonstrated efficacy in alleviating symptoms related to hormonal imbalances and the expanding opportunities for personalized treatment approaches.
GnRH agonists are utilized to suppress the release of gonadotropins, thereby reducing the production of sex hormones such as estrogen and testosterone, which exacerbate AIP symptoms. These therapies, administered via injections or nasal sprays, offer flexibility in treatment delivery, thus contributing to the growth of the segment.
By End-Users
Based on type, the market is divided into hospitals & clinics, ambulatory surgical centers, and research centers. The hospitals & clinics segment captured the largest acute intermittent porphyria market share of 67.89% in 2023.
Hospitals & clinics have emerged as the central facilities for the diagnosis, treatment, and management of this rare metabolic disorder. These healthcare facilities offer specialized care units staffed by trained professionals who utilize advanced diagnostic technologies, thereby enabling accurate and timely diagnosis of AIP cases.
Due to rising awareness and increased diagnosis rates, hospitals and clinics are experiencing a growing number of patients seeking AIP-related healthcare services. Technological advancements enhance their capabilities to provide personalized treatment plans tailored to individual patient needs.
Acute Intermittent Porphyria Market Regional Analysis
Based on region, the global market is classified into North America, Europe, Asia-Pacific, MEA, and Latin America.
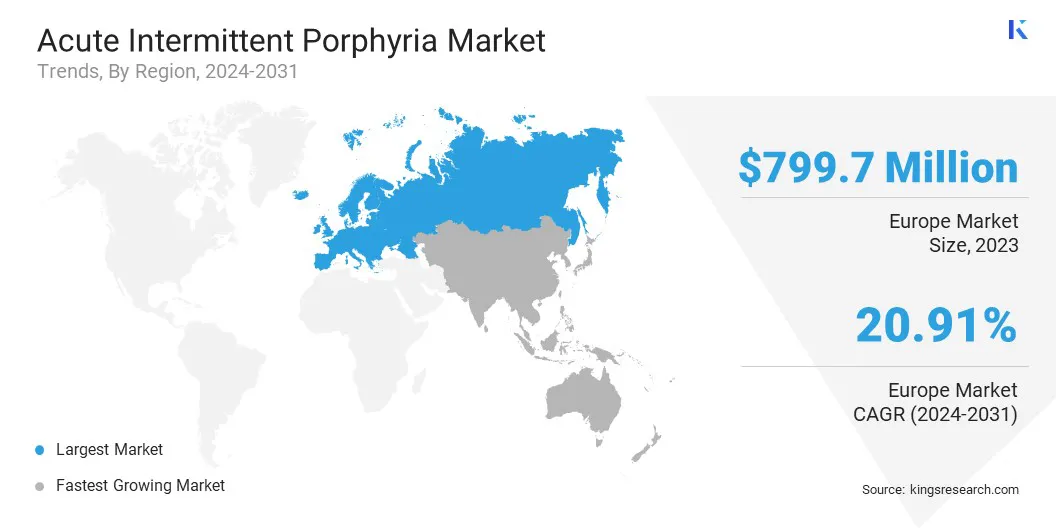
The Europe Acute Intermittent Porphyria Market share stood around 41.34% in 2023 in the global market, with a valuation of USD 799.7 million. This notable growth is fueled by the region's robust healthcare infrastructure, advanced medical research capabilities, and a high prevalence of metabolic disorders. The aging population and genetic predispositions contribute to the rising incidence of AIP in the region, highlighting the need for comprehensive management strategies.
- According to the American Porphyria Foundation, the estimated prevalence of AIP in Europe is approximately 5.9 per million people in the general population.
Europe's well-developed healthcare systems, coupled with access to advanced diagnostic and treatment facilities, facilitate timely diagnosis and effective management of AIP cases.
Asia-Pacific is anticipated to witness the fastest growth, depicting a CAGR of 22.12% over the forecast period. The growing incidence of metabolic disorders and rapidly improving healthcare infrastructure foster regional market expansion.
Rapid economic development has led to an expansion of access to diagnostic facilities and specialized treatment centers, which facilitat early diagnosis and comprehensive care for AIP patients. Additionally, increasing investments in medical research and innovation across the region are spurring efforts to uncover disease mechanisms, develop novel therapies, and enhance patient care protocols.
Competitive Landscape
The acute intermittent porphyria market report will provide valuable insight with an emphasis on the fragmented nature of the industry. Prominent players are focusing on several key business strategies such as partnerships, mergers and acquisitions, product innovations, and joint ventures to expand their product portfolio and increase their market shares across different regions.
Companies are implementing impactful strategic initiatives, such as expanding services, investing in research and development (R&D), establishing new service delivery centers, and optimizing their service delivery processes, which are likely to create new opportunities for market growth.
List of Key Companies in Acute Intermittent Porphyria Market
- Alnylam Pharmaceuticals, Inc.
- Recordati Rare Diseases
- Octapharma AG
- Clinuvel Pharmaceuticals
- Shandong octagon chemicals limited
- Glenmark Pharmaceuticals Ltd
- Intas Pharmaceutical Ltd.
- Sanofi
- Sun Pharmaceuticals Industries Ltd.
- Abbott Healthcare Pvt. Ltd.
The global acute intermittent porphyria market is segmented as:
By Treatment
- Gonadotropin-Releasing Hormone Analogues
- Prophylactic Hematin Infusions
By End-Users
- Hospitals & Clinics
- Ambulatory Surgical Centres
- Research Centers
By Region
- North America
- Europe
- France
- U.K.
- Spain
- Germany
- Italy
- Russia
- Rest of Europe
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Rest of Asia-Pacific
- Middle East & Africa
- GCC
- North Africa
- South Africa
- Rest of Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America


